FDA approves food processing aid to help eliminate Listeria
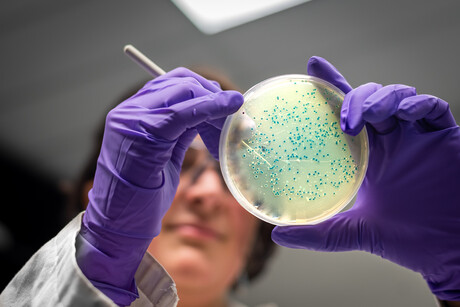
The United States (US) Food and Drug Administration (FDA) and the US Department of Agriculture (USDA) have granted GRAS (Generally Recognised as Safe) status to INNEO, a food processing aid designed to help eliminate Listeria monocytogenes. The technology, developed by Canadian biotechnology company Innodal, uses antimicrobial peptides to help eliminate the pathogen. With this approval, the company is set to launch industrial-scale projects with US processing companies.
The flagship product has been commercialised in Canada for four years and will now be available to the US market.
Designed to target Listeria monocytogenes with its antimicrobial action, the product aims to enhance the safety of ready-to-eat foods. The clean label solution is designed to preserve the taste, colour and texture of food. As a recognised processing aid, INNEO can be used by food processors without requiring ingredient label updates.
Laurent Dallaire, CEO and Co-Founder at Innodal, said: “Achieving GRAS status for INNEO reinforces our commitment to innovation and food safety. This official recognition now allows American food processors to use Inneo in full compliance with FDA regulations, opening new possibilities. We are convinced that this new step will contribute to improving the quality and safety of food products globally.”
Argon & Co partners with QAD | Redzone
Management consultancy Argon & Co partners with QAD | Redzone workforce technology to help...
Are consumers ready for food made from seafood sidestreams?
A European survey says 74% have a positive or neutral attitude towards eating food made from...
Novel shock method for calculating coffee flavour
US scientists have developed a quick test that applies voltage to black coffee to determine its...











