The use of Radial Flow Column (RFC) for large-scale chromatography processes
By Michael Feische, Head of R&D, Albert Handtmann Armaturenfabrik GmbH & Co. KG
Friday, 01 July, 2022

Huge market growth
Lactoferrin (LF) is found in the milk of all mammalians, also in milk and whey derived from cow milk. It is an 80 kDa iron-binding protein and represents about 0.3% of the total protein content of milk, which equals 60–200 mg/L. The thermal stability is 55°C.
Lactoferrin is a very powerful protein: it has antibacterial, antiviral, antifungal, anti-tumor, anti-inflammatory and anti-allergic effects. Isolated from cow’s milk, it is therefore a sought-after additive for foods such as functional food or sports nutrition. The use of lactoferrin has exploded in recent years, particularly as a supplement for baby nutrition.
Column chromatography
In chemistry, chromatography refers to all those physico-chemical separation processes in which the separation process is based on the distribution of a substance between a mobile and a stationary phase. Different substances of a sample are retained to different degrees by the stationary phase, while the mobile phase takes over the transport. Chromatographic analysis methods can include the following: classical column chromatography, high-performance liquid chromatography (HPLC) or ion exchange chromatography (IEX) as a special form of HPLC.
Fast protein liquid chromatography (FPLC) is a form of high-performance liquid chromatography that is often used for the analysis or purification of protein mixtures. In FPLC, the mobile phase is an aqueous solution or ‘buffer’. The buffer flow rate is controlled by a positive displacement pump and maintained at a constant flow rate. For equilibration and elution, substance-specific buffers are required. To adapt the elution to the substance to be separated, the buffer composition can be varied by dosing the individual components from external containers. The stationary phase is usually a spherical resin, which for example consists of cross-linked agarose or polymethacrylates, packed in a cylindrical stainless steel column. FPLC resins are available in a wide range of particle sizes and surface modifications, depending on the application.
In ion exchange chromatography, substances can be separated according to their charge. It is based on the formation of heteropolar bonds between the matrix and the mobile phase, whereby the desired charged protein binds. The elution can be done by means of gradients. As soon as the charge binding between eluent and protein is higher than between matrix and protein, the protein migrates into the solution. By detecting the flow at 280 nm, any passing protein can be detected as a peak. In lactoferrin recovery, LF is obtained by ion exchange chromatography.
What is radial flow?
The radial flow stands for a continuous flow and high flow rate through the entire adsorber (24/7), a low differential pressure at a high flow rate, the processing of large feed volumes and a good scalability by increasing the separation distance of the corresponding radial flow column.
More advantages of RFC are the following:
- Bidirectional flow
- Ability to sterilise and autoclave the column
- Availability of different screen sizes
- Small footprint
- Low dead space volume
- Minimum P
- Robust design
- Specific packing station for easy packing and unpacking, together with minimizing chromatographic resin losses
The standard column volume (CV) is 240 and 260 litres respectively at a separation distance of 13.5 cm. In principle, up to 2000 litres of resin volume per column are possible. For beer applications, this will allow processing flow rate of ~ 22,000 litres per hour, for milk applications ~15,000 litres per hour.
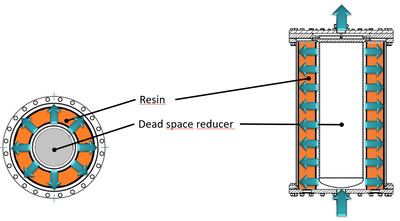
Axial vs. radial flow column
While an axial flow column design offers the advantage of a higher flexibility in the separation distance, the radial flow column has much more to offer. Below are the specifications for a radial flow column, which make a radial flow column superior to an axial flow column:
- Reduced pressure drop of 20–50 %
- Improved loading efficacy
- Reduced feed handling complexity
- High binding kinetics (dynamic binding capacity)
- High process robustness (larger process tolerance window)
- Higher throughput
- Higher efficiency
- Higher purity

Is it possible to convert existing processes from axial to radial flow columns?
A conversion from an axial to a radial flow column is possible as long as the required separation distance for the efficient separation is max. 13.5 cm, which is in most of the ion-exchange based chromatography processes the case. The change from axial to radial can be justified as described above: radial flow columns have significantly lower pressures than axial columns. This means that the column can be operated at a higher flow rate, resulting in a higher throughput and, in relation to time, a higher turnover.
How to extract lactoferrin from milk
For the industrial extraction of the protein lactoferrin from milk and whey, radial flow columns are used. The whole milk is collected at dairy farms and stored in refrigerated conditions until quality test passed and transported to a dairy processing facility. Once the milk arrives at the dairy facility, it undergoes cream separation to remove fat and collect skim milk. Then the skim milk goes through a pasteurizer to kill pathogenic bacteria before any further process. For bovine lactoferrin (bLF) extraction, the ideal option is to extract the lactoferrin before the pasteurizer process step. In such cases, skim milk is pumped through a chromatography column packed with a suitable resin (strong ion exchanger) until the maximum binding capacity of the resin has been reached. The column is then washed with water and low salt buffers, before the bovine lactoferrin is eluted by applying a buffer with high salt solution. Afterwards the resin in the column is regenerated for next purification cycle by a CIP procedure using sodium hydroxid (NaOH). The purified lactoferrin-/salt solution is further processed to remove the salt and concentrate the solution by diafiltration (DF) and ultrafiltration (UF). LF is further concentrated using a second UF process step and then dried using freeze-drying or spray drying technologies. The powder is formulated and packed to a desired pack size.

Applications of column chromatography
In addition to lactoferrin extraction, the radial flow column is also used in the food industry and in large-scale chromatography processes. Here are just a few areas of application:
- Bitter substances from citrus juices
- Polyphenol reduction in beer
- Taurine extraction
- Extraction of potato protein
- Taste and smell removal from collagen
- Removal of riboflavin from whey
- Milk and whey protein purification
Enhancing Food Quality and Safety with METTLER TOLEDO
This article explores key testing methods including moisture analysis, pH measurement, titration,...
X-ray Food Inspection on the Road: Safety on Wheels!
Australian food manufacturers are being offered a rare, hands-on opportunity to evaluate X-ray...
Rising costs push food processors to switch to more agile inventory management
Cost and growth pressures are pushing manufacturers to rethink ERP and adopt more agile and...











